Traditional control and task automation have been successfully demonstrated in a variety of structured, controlled environments through the use of highly specialized modeled robotic systems in conjunction with multiple sensors. However, application of autonomy in endoscopic surgery is very challenging, particularly in soft tissue work, due to the lack of high-quality images and the unpredictable, constantly deforming environment. In this work, we propose a novel surgical perception framework, SuPer, for surgical robotic control. This framework continuously collects 3D geometric information that allows for mapping of a deformable surgical field while tracking rigid instruments within the field. To achieve this, a model-based tracker is employed to localize the surgical tool with a kinematic prior in conjunction with a model-free tracker to reconstruct the deformable environment and provide an estimated point cloud as a mapping of the environment. The proposed framework was implemented on the da Vinci Surgical System in real-time with an end-effector controller where the target configurations are set and regulated through the framework. Our proposed framework successfully completed autonomous soft tissue manipulation tasks with high accuracy. The demonstration of this novel framework is promising for the future of surgical autonomy. In addition, we provide our dataset for further surgical research.
SuPer Deep: A Surgical Perception Framework for Robotic Tissue Manipulation using Deep Learning
Robotic automation in surgery requires precise tracking of surgical tools and mapping of deformable tissue. Previous works on surgical perception frameworks require significant effort in developing features for surgical tool and tissue tracking. In this work, we overcome the challenge by exploiting deep learning methods for surgical perception. We integrated deep neural networks, capable of efficient feature extraction, into the tissue reconstruction and instrument pose estimation processes. By leveraging transfer learning, the deep learning based approach requires minimal training data and reduced feature engineering efforts to fully perceive a surgical scene. The framework was tested on three publicly available datasets, which use the da Vinci Surgical System, for comprehensive analysis. Experimental results show that our framework achieves state-of-the-art tracking performance in a surgical environment by utilizing deep learning for feature extraction.
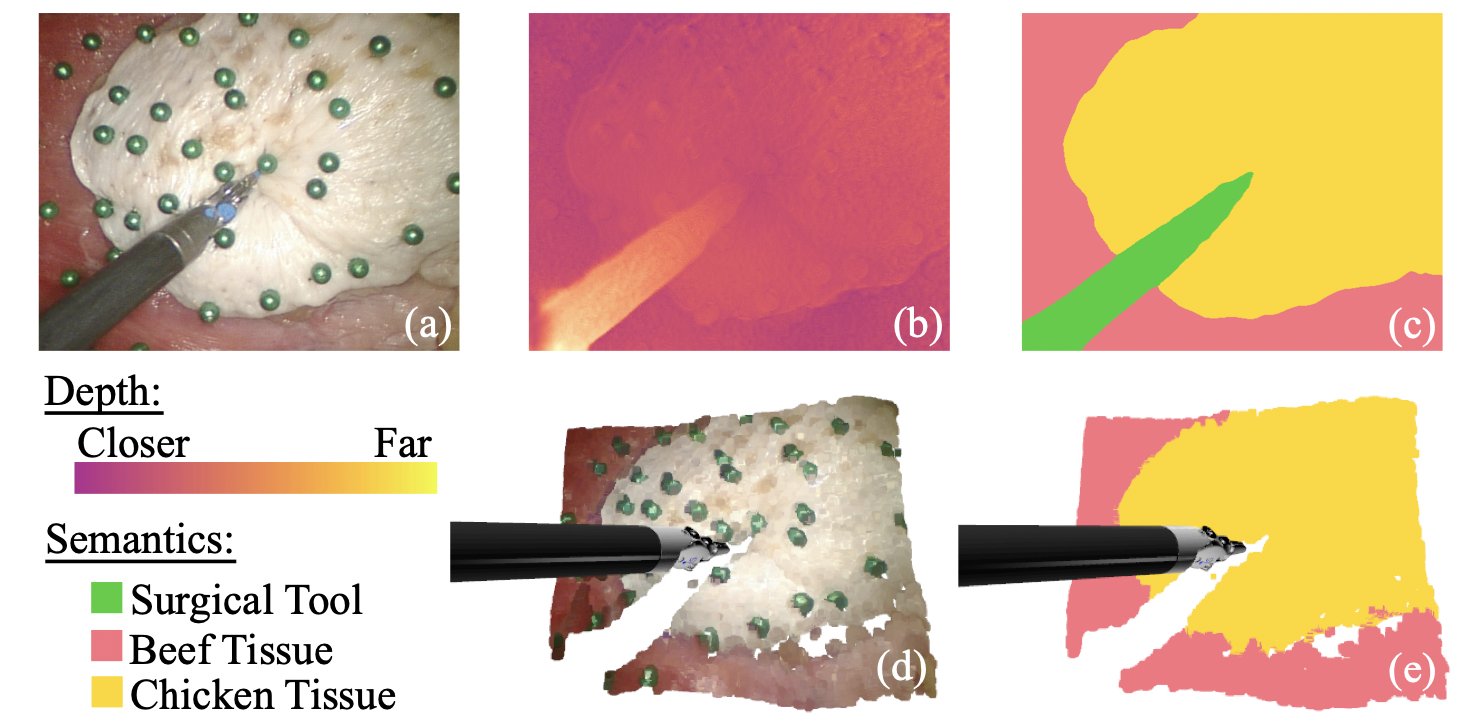
Semantic-SuPer: A Semantic-aware Surgical Perception Framework for Endoscopic Tissue Identification, Reconstruction, and Tracking
Accurate and robust tracking and reconstruction of the surgical scene is a critical enabling technology toward autonomous robotic surgery. Existing algorithms for 3D perception in surgery mainly rely on geometric information, while we propose to also leverage semantic information inferred from the endoscopic video using image segmentation algorithms. In this paper, we present a novel, comprehensive surgical perception framework, Semantic-SuPer, that integrates geometric and semantic information to facilitate data association, 3D reconstruction, and tracking of endoscopic scenes, benefiting downstream tasks like surgical navigation. The proposed framework is demonstrated on challenging endoscopic data with deforming tissue, showing its advantages over our baseline and several other state-of the-art approaches.
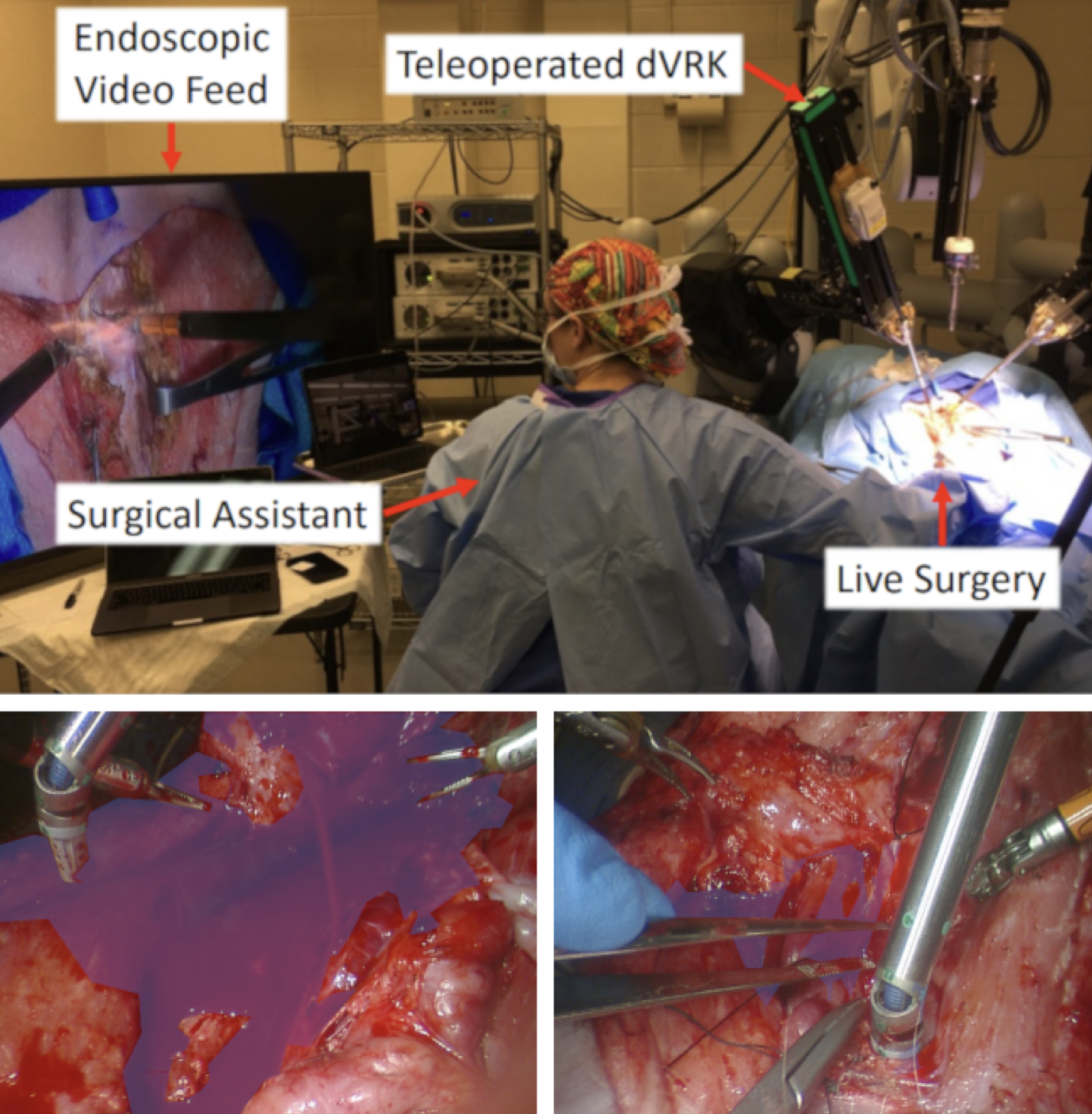
Hemorrhaging occurs in surgeries of all types, forcing surgeons to quickly adapt to the visual interference that results from blood rapidly filling the surgical field. Introducing automation into the crucial surgical task of hemostasis management would offload mental and physical tasks from the surgeon and surgical assistants while simultaneously increasing the efficiency and safety of the operation. The first step in automation of hemostasis management is detection of blood in the surgical field. To propel the development of blood detection algorithms in surgeries, we present HemoSet, the first blood segmentation dataset based on bleeding during a live animal robotic surgery. Our dataset features vessel hemorrhage scenarios where turbulent flow leads to abnormal pooling geometries in surgical fields. These pools are formed in conditions endemic to surgical procedures -- uneven heterogeneous tissue, under glossy lighting conditions and rapid tool movement. We benchmark several state-of-the-art segmentation models and provide insight into the difficulties specific to blood detection. We intend for HemoSet to spur development of autonomous blood suction tools by providing a platform for training and refining blood segmentation models, addressing the precision needed for such robotics.