Autonomous robotic surgery has seen significant progression over the last decade with the aims of reducing surgeon fatigue, improving procedural consistency, and perhaps one day take over surgery itself. However, automation has not been applied to the critical surgical task of controlling tissue and blood vessel bleeding--known as hemostasis. The task of hemostasis covers a spectrum of bleeding sources and a range of blood velocity, trajectory, and volume. In an extreme case, an un-controlled blood vessel fills the surgical field with flowing blood. In this work, we present the first, automated solution for hemostasis through development of a novel probabilistic blood flow detection algorithm and a trajectory generation technique that guides autonomous suction tools towards pooling blood. The blood flow detection algorithm is tested in both simulated scenes and in a real-life trauma scenario involving a hemorrhage that occurred during thyroidectomy. The complete solution is tested in a physical lab setting with the da Vinci Research Kit (dVRK) and a simulated surgical cavity for blood to flow through. The results show that our automated solution has accurate detection, a fast reaction time, and effective removal of the flowing blood. Therefore, the proposed methods are powerful tools to clearing the surgical field which can be followed by either a surgeon or future robotic automation developments to close the vessel rupture.
Bimanual Regrasping for Suture Needles using Reinforcement Learning for Rapid Motion Planning
Regrasping a suture needle is an important process in suturing, and previous study has shown that it takes on average 7.4s before the needle is thrown again. To bring efficiency into suturing, prior work either designs a task-specific mechanism or guides the gripper toward some specific pick-up point for proper grasping of a needle. Yet, these methods are usually not deployable when the working space is changed. These prior efforts highlight the need for more efficient regrasping and more generalizability of a proposed method. Therefore, in this work, we present rapid trajectory generation for bimanual needle regrasping via reinforcement learning (RL). Demonstrations from a sampling-based motion planning algorithm is incorporated to speed up the learning. In addition, we propose the ego-centric state and action spaces for this bimanual planning problem, where the reference frames are on the end-effectors instead of some fixed frame. Thus, the learned policy can be directly applied to any robot configuration and even to different robot arms. Our experiments in simulation show that the success rate of a single pass is 97%, and the planning time is 0.0212s on average, which outperforms other widely used motion planning algorithms. For the real-world experiments, the success rate is 73.3% if the needle pose is reconstructed from an RGB image, with a planning time of 0.0846s and a run time of 5.1454s. If the needle pose is known beforehand, the success rate becomes 90.5%, with a planning time of 0.0807s and a run time of 2.8801s.
Robotic Autonomy in Endocrine surgery
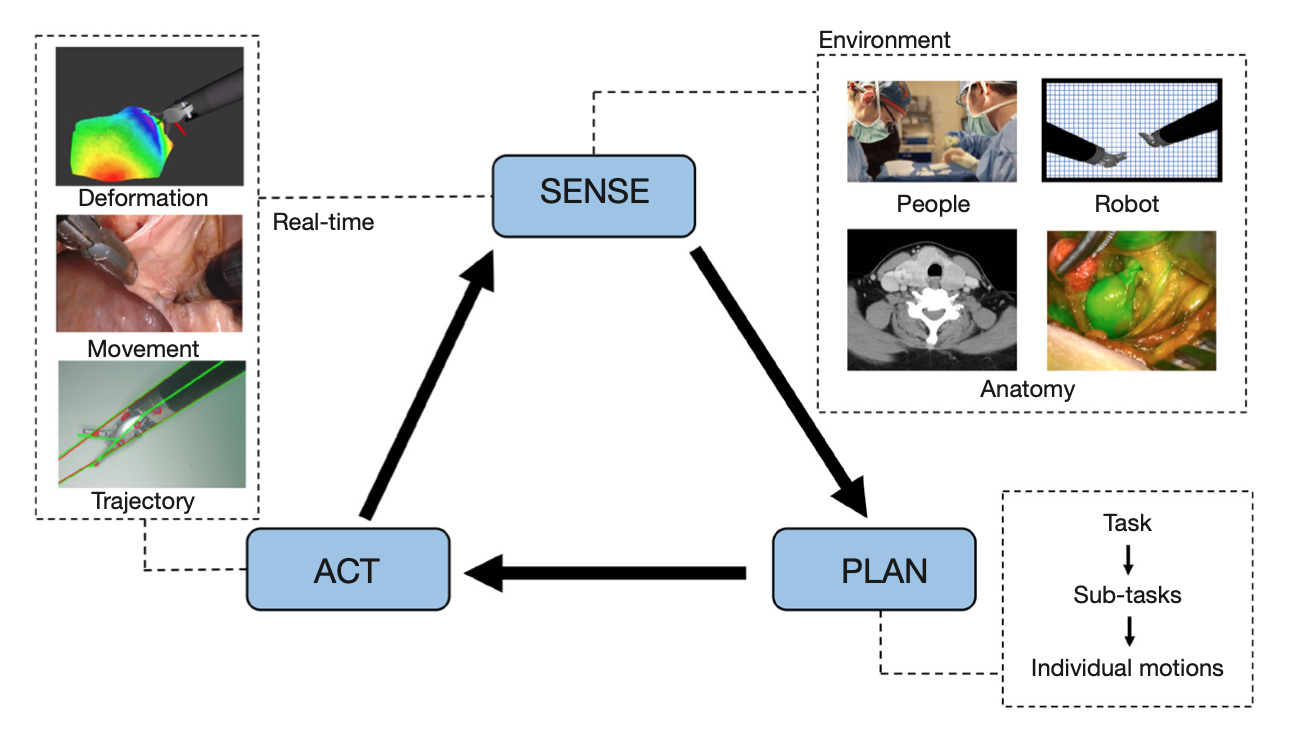
An autonomous robot is able to perceive its environment, make decisions and plans, then execute multi-step functions. Although surgical robotic technologies are widely accepted, robotic autonomy has not made its way into the operating rooms of today. As a result, robotic automation is an unfamiliar topic for most surgeons. In this review, we define the spectrum of robotic autonomy, provide a review of key components required for autonomy, and discuss some of the ways in which autonomy may be applied to endocrine surgery in the future. The integration of advanced imaging and surgical environment perception could augment surgical visualization and lead to the development of advanced semi-autonomous and autonomous robotic algorithms. The hope is that advanced robotic surgical technologies can make robotic surgeries safer, more efficient, precise and allow for greater patient access to advanced surgical expertise.
Chronic wounds, including diabetic ulcers, pressure ulcers, and ulcers secondary to venous hypertension, affects more than 6.5 million patients and a yearly cost of more than $25 billion in the United States alone. Chronic wound treatment is currently a manual process, and we envision a future where robotics and automation will aid in this treatment to reduce cost and improve patient care. In this work, we present the development of the first robotic system for wound dressing removal which is reported to be the worst aspect of living with chronic wounds. Our method leverages differentiable physics-based simulation to perform gradient-based Model Predictive Control (MPC) for optimized trajectory planning. By integrating fracture mechanics of adhesion, we are able to model the peeling effect inherent to dressing adhesion. The system is further guided by carefully designed objective functions that promote both efficient and safe control, reducing the risk of tissue damage. We validated the efficacy of our approach through a series of experiments conducted on both synthetic skin phantoms and real human subjects. Our results demonstrate the system's ability to achieve precise and safe dressing removal trajectories, offering a promising solution for automating this essential healthcare procedure.